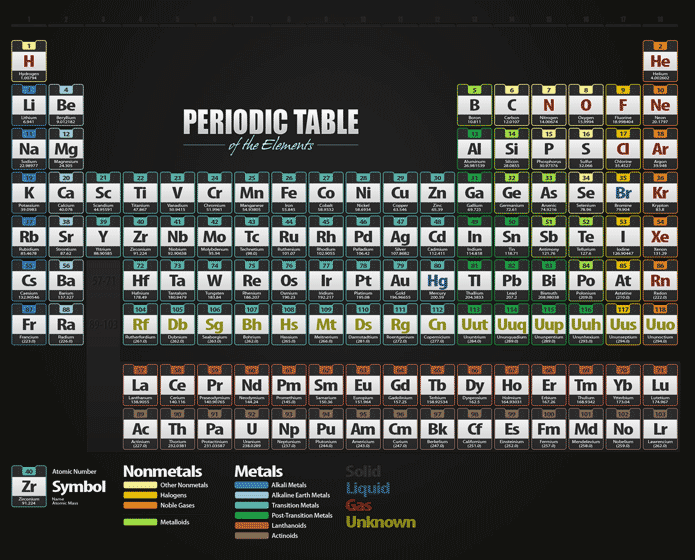
So according to the Royal Society of Chemistry, Mendeleev wrote the properties of each element on cards, and then he started ordering them by increasing atomic weight. But they weren't enough to usefully sort the 55 additional chemical elements known at the time. Gordin in his book "A Well-Ordered Thing: Dmitrii Mendeleev and the Shadow of the Periodic Table" (Princeton University Press, Revised Edition 2018). The first section of Mendeleev's book dealt with just eight of the known elements - carbon, hydrogen, oxygen, nitrogen, chlorine, fluorine, bromine and iodine - and those two strategies worked for those particular elements, according to Michael D. Just two strategies existed at the time to categorize these elements: separating them into metals and nonmetals or grouping them by an element's number of valence electrons (or those electrons in the outermost shell). At the time, there were 63 known chemical elements, each with an atomic weight calculated using Avogadro's hypothesis, which states that equal volumes of gases, when kept at the same temperature and pressure, hold the same number of molecules. Putting the elements in any kind of order would prove quite difficult. Valence electrons are the electrons of an atom that are highest in energy and in the outermost orbitals.(Image credit: Oxford Science Archive/Print Collector/Getty Images) The transition elements are metals that have a partially filled d subshell and comprise groups 3 through 12 and the lanthanides and actinides. The s-block is the section of elements on the periodic table composed of the first two groups or columns. The periodic law is the principle that the properties or characteristics of the elements recur periodically as their atomic numbers increase. PeriodĪ period is a horizontal row of the periodic table. The p-block is the section of the periodic table composed of the last six groups or columns. OxideĪn oxide is a compound composed of a metal and oxygen. The oxidation state is the charge associated with an atom. The term non-metals is used to classify the elements H, C, N, P, O, S, and Se. They are generally very stable, colorless, and odorless. MetalsĪ metal is a substance that conducts heat and electricity, is shiny, and can be hammered into sheets or drawn into a wire. They are the elements B, Si, Ge, As, Sb, Te, and Po. Malleability means an object is capable of being hammered into sheets or shapes. Groups 1-2 and 13-18 are called the main-group elements. Along with the actinides they are often called the f-elements because they have valence electrons in the f shell. The lanthanides comprise elements 57 through 71. The ionization energy is the energy needed to remove an electron from an atom. They comprise group 17 of the periodic table, from F through At. The halogen elements are a subset of the nonmetals. It is a column of elements in the periodic table that all have similar properties. Flame TestĪ flame test is a test for detecting the presence of certain metals by determining the color they emit when put in a flame. FamilyĪ taxon (group of organisms) made up of related genera. The f-block is the section of elements below the main section of the periodic table consisting of the lanthanide and actinide elements. ExothermicĮxothermic is the term used to describe a reaction that releases heat. ElementĪn element is a substance composed of atoms with identical atomic number. ElectronegativityĮlectronegativity is a measure of the attraction an atom has for electrons. 
Electron AffinityĮlectron affinity is the amount of energy absorbed when an electron is added to an atom. The d-block is the section of elements on the periodic table in groups 3 – 12. DiatomicĪ complex consisting of two atoms. ConductorĪ material that conducts heat or electricity, which means electrons can easily flow through the object. 
Chemical SymbolĪ chemical symbol is a notation of one or two letters representing a chemical element. The atomic weight is the average mass of an atom of an element. The atomic radius is the total distance from an atom's nucleus to the outermost electron orbital. Atomic OrbitalĪtomic orbitals are regions of space around the nucleus of an atom where an electron is most likely to be found. The atomic number is the number of protons in an atomic nucleus. The alkaline earth metals make up the second column (group) of the periodic table, from Be through Ra. The alkali metals make up the first column (group) of the periodic table, and comprise Li through Fr. They, along with the actinides, are often called the f-elements because they have valence electrons in the f shell. The actinides comprise elements 89 through 103.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
